Concept Review Section Covalent Bonds

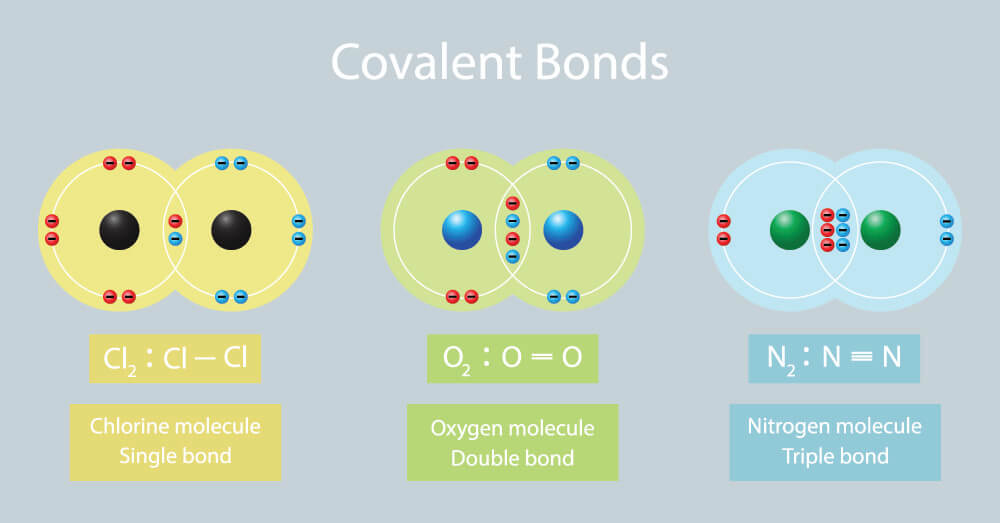
A covalent bond forms when two or more valence electrons are attracted by the positively charged nuclei of two atoms and thus are shared between both atoms.
Concept review section covalent bonds. There are strong bonds between the atoms in a piece of quartz these bonds give quartz a. The number of bonds an element forms in a covalent compound is determined by the number of electrons it needs to reach octet. Contrast ionic and covalent bonds.
Ionic and covalent bonding 1. A covalent bond forms when two or more valence electrons are attracted by the positively charged nuclei of two atoms and thus are shared between both atoms. A covalent bond forms when two or more valence electrons are attracted by the positively charged nuclei of two atoms and thus are shared between both atoms.
Compounds that involve a metal binding with either a non metal will display ionic bonding. But you may not obsession to influence or. 2.
Holt chemistry concept review answers chapter 13 the two types of bonds are ionic bonds and covalent bonds. In an ionic bond the atoms are bound together by the attraction between oppositely charged ions. Explain why atoms will often join together to form bonds.
Use the concept of potential energy to describe how a covalent bond forms between two atoms. 3. If the electron is shared equally between the atoms forming a covalent bond then the bond is said to be nonpolar.
As the atoms involved in the formation of a covalent bond approach each other the electron proton attraction is stronger than the electron electron and proton proton repulsions. Explain how it is possible. You can gain access to the books wherever you desire even you are in the bus office home and additional places.